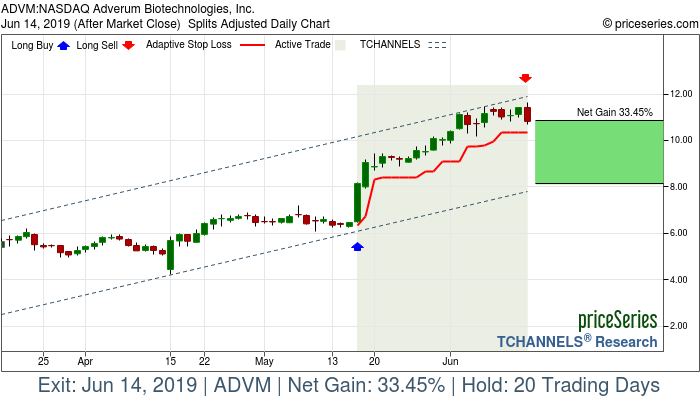
Avalanche Biotechnologies, Inc., a gene therapy company, engages in discovering and developing novel medicines that offer therapeutic benefit to patients suffering from chronic or debilitating disease. It primary focuses on developing AVA-101, which is in a Phase IIa trial for the treatment of wet age-related macular degeneration (AMD). The company is also developing AVA-201, a product candidate for the prevention of wet AMD; AVA-311 that is in preclinical studies for the treatment of juvenile X-linked retinoschisis, a rare genetic disease of the retina with no approved therapy; and AVA-322 and AVA-323 for the treatment of color vision deficiency. Avalanche Biotechnologies, Inc. has a collaboration agreement with Regeneron Pharmaceuticals, Inc. to research, develop, and commercialize gene therapy products. The company was founded in 2006 and is headquartered in Menlo Park, California.
Trade Type
| ReliabilityScore™
| Entry Date
| Entry Price
| Sell Date
| Sell Price
| Net Gain
| Hold Time
|